 The assignment of electronegativity values isīased on the affinity of a nucleus to accepting an electron. This phenomenon can also be explained through the principle ofĮlectronegativity. The right of the noble gases) H, Li, Na, and K tend to exhibit high Left of the noble gases) F, Cl, and Br, and the alkali metals (to The alkali metals, found in group 1 of the periodic table (formally known as group IA), are so reactive that they are generally found in nature combined with other elements. Table since these are just one electron away from the noble gasĬonfiguration in the electron shells. Will be those directly adjacent to the noble gases on the periodic Pure argon gas to avoid oxidation that might occur from atmosphericĪir because argon is generally unreactive.įrom this observation, it follows that the most reactive species It is a member of the chalcogen group on the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as well as with other compounds. Many experiments are carried out in vacuum under Oxygen is a colourless, odourless reactive gas, the chemical element of atomic number 8 and the life-supporting component of the air. Both groups either need to gain or lose an electron, which is relatively easy, so they react more easily. 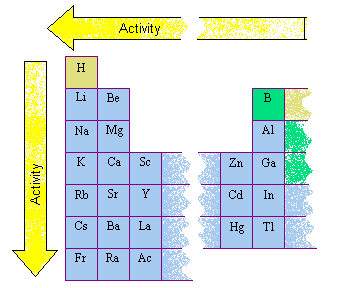
Lithium, sodium, and potassium all react with water, for example. This is due in part to their outer valence shells. The elements toward the bottom left corner of the periodic table are the metals that are the most active in the sense of being the most reactive. Species are very stable and accordingly relatively inert toĬhemical reaction. GCSE CCEA The periodic table - (CCEA) Group 1 (I) reactivity The periodic table helps to categorise the known elements and make predictions about ones that we haven’t yet discovered. Explanation: Although a teacher may say one is more reactive than the others, the reality is that both are equally reactive. This corresponds to the lastĬolumn of the periodic table, known as the "noble gases." These In fact solid sodium will even explode in waterGroup 1. But the alkali metals are just as reactive. On descending a group in the periodic table, on average the outermost electrons get further and further away from the nucleus and as a result, become easier to remove from the atom. Really, the most reactive elements on the Periodic Table tend to be found in the Halogen Group. /caesium-cs--chemical-element--3d-rendering-isolated-on-black-background-922704282-5adb6de08e1b6e0037170151.jpg)
The least reactive elements are those who have a full outermost valence shell ie they have 8 electrons in the outer shell so elements such as helium, neon, radon or the transition elements. Whats more, to the surprise of most chemists and going against the well-known trends of the periodic table, it turns out that francium is not the most reactive metal. 
Orbital theory suggests that the most stable species are those The reactivity of an element can be determined by looking at the electron configuration of the element. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
